Kelly Warner Law Firm Blames USA…
5/17/17 Based on the information released in The Washington Post article…
By – USA HeraldAaron Kelly Law Firm Resorts To…
Professor Volokh thereafter filed a bar complaint against Dan Warner…
By – Jeff WattersonArizona Bar Opens Investigation on Attorney…
USA Herald recently reported on a developing story involving Attorneys…
By – Paul O'NealJosh Duggar Appeal Denied as Convicted…
Josh Duggar Transfer to Federal Medical Center in Fort Worth…
By – Jackie AllenFederal Judge Lets ’86 47′ Flag…
“Use of the statement ’86 47′ and other variations will…
By – Samuel LopezSabrina Carpenter Granted Restraining Order Following…
Citing “severe emotional distress,” the American pop star has successfully…
By – Tyler BrooksThe Diddy Fallout: Cassie Fights Back…
Two Diverging Paths These latest developments highlight an irreversible divide.…
By – Tyler BrooksSouth Carolina Jury Clears Store Owner…
A South Carolina courtroom erupted with emotion Monday after a…
By – Tyler BrooksArcher Aviation: The eVTOL Takeoff Facing…
Conclusion: A Bimodal Investment Archer Aviation is not failing operationally;…
By – Tyler BrooksSleeping Dog Documentary Chronicles Jeremy Corbell’s…
The film reportedly features material that later appeared in recently…
By – Jackie AllenKendall Jenner, Jacob Elordi and the…
Think about the kind of relationships this family was associated…
By – Nathan KayChaotic Midnight Shooting Leaves 3 Bloodied…
Gunman Remains Loose as Motive Eludes Detectives Law enforcement officials…
By – Tyler Brooks43-year-old Man Hospitalized After a Stranger…
Investigators Collect Critical Ballistic Evidence Crime scene investigators spent several…
By – Tyler BrooksHurricane Season Starts Today – Here’s…
Texas Faces Reduced But Dangerous Landfall Risk While the federal…
By – Tyler BrooksU.S. Military Strike In The Eastern…
“The premeditated and intentional killings lack any plausible legal justification.”…
By – Tyler BrooksRare Blue Micromoon Lights Up the…
The Moon’s changing apparent size is simply a consequence of…
By – Jackie AllenMurder-for-hire Ends with Life Sentence for…
Defense Argued Lack of Evidence in Murder-for-Hire Plot Defense attorneys…
By – Jackie AllenFrank Lloyd Wright and the Taliesin…
The Taliesin Murders In August 1914, Taliesin became the scene…
By – Jackie AllenHollywood at a Crossroads: Spencer Pratt…
Spencer Pratt Presents a Competing Vision for Recovery While…
By – Jackie AllenHayden Panettiere has a Memoir About…
Rise to Fame on Heroes and Nashville Panettiere’s breakthrough came…
By – Jackie AllenBlue Origin Rocket Explodes in Massive…
Satellite Launch Mission Now in Doubt Before the explosion, the…
By – Jackie AllenBlue Origin Rocket Explodes in Massive…
Satellite Launch Mission Now in Doubt Before the explosion, the…
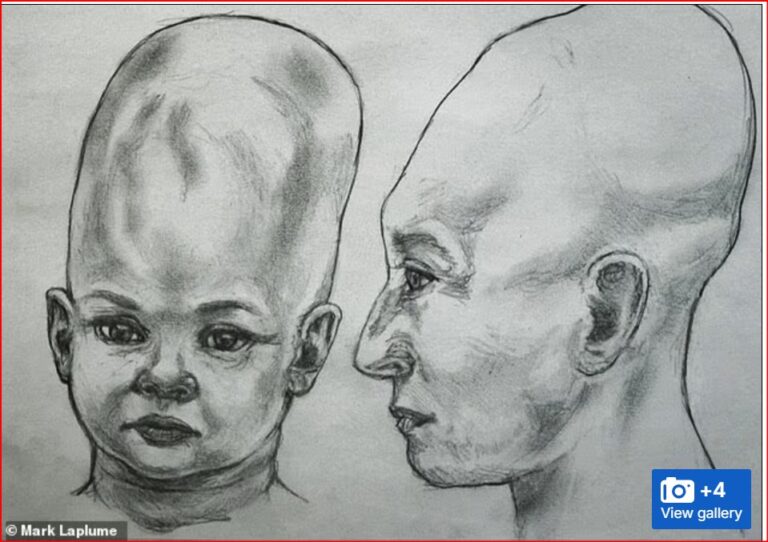
By – Jackie AllenAlien Coneheads: New DNA Study Doesn’t…
As a result, the study could not determine the individuals’…
By – Jackie AllenTrump’s Alien.gov Reveal Turns Into Immigration…
A Dangerous Game with Public Trust The larger issue may…
By – Samuel LopezTrump’s UFO files reveal mysterious flying…
Information about NASA’s moon missions is available through NASA Apollo…
By – Jackie AllenWho’s Lying? E. Jean Carroll Faces…
Todd Blanche Recused from E. Jean Carroll Matter One notable…
By – Jackie AllenSuper El Niño: Will 2026 be…
Professor Bill McGuire warned that temperatures in some parts of…
By – Jackie AllenArcher Aviation: The eVTOL Takeoff Facing…
Conclusion: A Bimodal Investment Archer Aviation is not failing operationally;…
By – Tyler BrooksFrom a Casual Night Out to…
It Doesn’t Happen Here’: Quiet Suburb Left Shattered After Fatal…
By – Tyler BrooksFrom Folklore to High Finance: The…
Thematic ETFs: Investment funds capturing capital through aerospace stock aggregation.…
By – Tyler BrooksAnthropic Files Historic IPO Triggering Fierce…
Anthropic Files Historic IPO Triggering Fierce Wall Street Ethics War…
By – Tyler BrooksManhunt underway for Florida felon Adriel…
Aggravated battery on a pregnant victim Multiple counts of domestic…
By – Tyler BrooksHawaii Warns Communities of Impending Kilauea…
Dangerous Pollutants Drift Across Regions Massive clouds of water vapor,…
By – Tyler BrooksNew Pill Doubles Survival for Pancreatic…
Federal Agencies Rush Public Patient Access The dramatic clinical success…
By – Tyler BrooksFDA warns public as cookie firm…
“The FDA is issuing this alert to ensure customers are…
By – Tyler BrooksTrump orders CDC to slash childhood…
Pediatricians Defy New Rules as Legal Battle Escalates The administration’s…
By – Tyler BrooksUSDA warns Americans over Salmonella in…
Federal Inspectors Order Kitchen Cleanout Federal safety teams are urging…
By – Tyler BrooksGKN Aerospace’s Biggest Battle May Not…
Commercial general liability policies often contain exclusions relating to pollutants,…
By – Samuel LopezGarden Grove Chemical Crisis Sparks Class…
By Samuel López | USA Herald A full-scale legal and…
By – Samuel LopezWembanyama in Tears: Spurs Dethrone Thunder…
Victor Wembanyama: 22 points, controlling the paint defensively Julian Champagnie:…
By – Tyler BrooksSupreme Court signals 27 states could…
Timeline of the Historic Legal Battles The path to the…
By – Tyler BrooksMauricio Pochettino sounds alarm on Chris…
Mauricio Pochettino sounds alarm on Chris Richards injury By Tylor…
By – Tyler BrooksUSMNT star Chris Richards tears two…
The countdown to the 2026 World Cup begins The United…
By – Tyler Brooks“Money” Mayweather Tucks Tail: $100 Million…
Mayweather’s representatives responded more carefully, stating that the boxer is…
By – Samuel LopezMackenzie Shirilla Sent Text Messages to…
Police later concluded there was no evidence indicating she had…
By – Jackie AllenNo posts found.
No posts found.