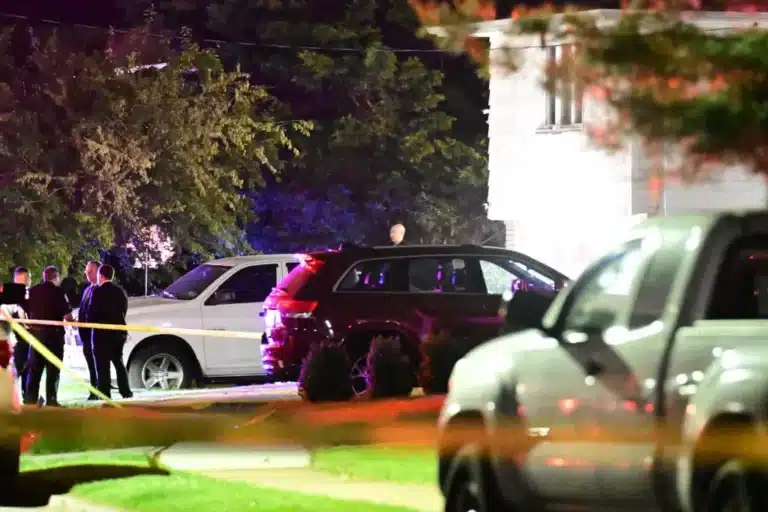Kelly Warner Law Firm Blames USA…
In what appears as a desperate attempt to defend multiple…
By – USA HeraldAaron Kelly Law Firm Resorts To…
Attorney Aaron Kelly and his law partner Daniel Warner are…
By – Jeff WattersonArizona Bar Opens Investigation on Attorney…
USA Herald recently reported on a developing story involving Attorneys…
By – Paul O'NealJosh Duggar Appeal Denied as Convicted…
Josh Duggar remains behind bars after a federal judge denied…
By – Jackie AllenFederal Judge Lets ’86 47′ Flag…
An Obama-appointed judge just ruled a political group can keep…
By – Samuel LopezSabrina Carpenter Granted Restraining Order Following…
Citing “severe emotional distress,” the American pop star has successfully…
By – Tyler BrooksThe Diddy Fallout: Cassie Fights Back…
As Sean “Diddy” Combs serves time behind bars, the shockwaves…
By – Tyler BrooksSouth Carolina Jury Clears Store Owner…
A South Carolina courtroom erupted with emotion Monday after a…
By – Tyler BrooksArcher Aviation: The eVTOL Takeoff Facing…
Strategic Analysis — June 2026 The electric vertical takeoff and…
By – Tyler BrooksSleeping Dog Documentary Chronicles Jeremy Corbell’s…
The new documentary Sleeping Dog arrives at a pivotal moment…
By – Jackie AllenKendall Jenner, Jacob Elordi and the…
I’ve been writing about royals and celebrities for 20 years.…
By – Nathan KayChaotic Midnight Shooting Leaves 3 Bloodied…
Downtown San Jose gunfire wounds 3, sparks wild building crash…
By – Tyler Brooks43-year-old Man Hospitalized After a Stranger…
Stranger shoots San Antonio man, 43, through door By Tyler…
By – Tyler BrooksHurricane Season Starts Today – Here’s…
Texas faces 20% hurricane risk as season begins By Tyler…
By – Tyler BrooksU.S. Military Strike In The Eastern…
U.S. Pacific boat strike kills 3, casualties cross 200 By…
By – Tyler BrooksRare Blue Micromoon Lights Up the…
Skywatchers are in for a unique celestial event as a…
By – Jackie AllenMurder-for-hire Ends with Life Sentence for…
A shocking Murder-for-hire case that spanned multiple states has concluded…
By – Jackie AllenFrank Lloyd Wright and the Taliesin…
In The Killer and Frank Lloyd Wright, veteran true-crime author…
By – Jackie AllenHollywood at a Crossroads: Spencer Pratt…
Los Angeles has its primary election this Tuesday, and the…
By – Jackie AllenHayden Panettiere has a Memoir About…
Hayden Panettiere is revealing the emotional toll of growing up…
By – Jackie AllenBlue Origin Rocket Explodes in Massive…
Blue Origin suffered a major setback Thursday night when one…
By – Jackie AllenBlue Origin Rocket Explodes in Massive…
Blue Origin suffered a major setback Thursday night when one…
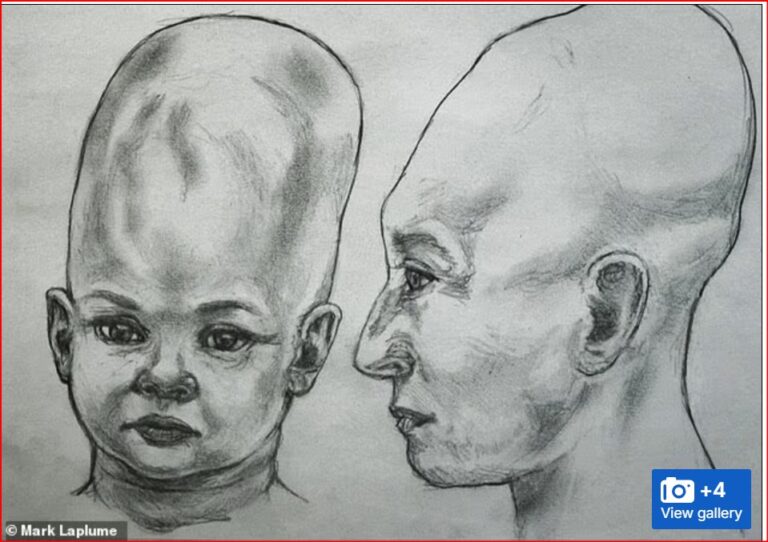
By – Jackie AllenAlien Coneheads: New DNA Study Doesn’t…
The mystery surrounding the so-called Alien Coneheads of Peru has…
By – Jackie AllenTrump’s Alien.gov Reveal Turns Into Immigration…
INSIDE THIS REPORT What millions thought would be a historic…
By – Samuel LopezTrump’s UFO files reveal mysterious flying…
The newly released UFO Files from the Trump administration have…
By – Jackie AllenWho’s Lying? E. Jean Carroll Faces…
Author and columnist E. Jean Carroll is once again at…
By – Jackie AllenSuper El Niño: Will 2026 be…
Scientists across the globe are increasingly warning that a potential…
By – Jackie AllenArcher Aviation: The eVTOL Takeoff Facing…
Strategic Analysis — June 2026 The electric vertical takeoff and…
By – Tyler BrooksFrom a Casual Night Out to…
It Doesn’t Happen Here’: Quiet Suburb Left Shattered After Fatal…
By – Tyler BrooksFrom Folklore to High Finance: The…
Wall Street and Global Powers Monetize UFO Craze By Tyler…
By – Tyler BrooksAnthropic Files Historic IPO Triggering Fierce…
Anthropic Files Historic IPO Triggering Fierce Wall Street Ethics War…
By – Tyler BrooksManhunt underway for Florida felon Adriel…
Manhunt underway for Florida felon Adriel Martinez after release breach…
By – Tyler BrooksHawaii Warns Communities of Impending Kilauea…
Hawaii Warns Communities of Impending Kilauea Ashfall By Tyler Brooks…
By – Tyler BrooksNew Pill Doubles Survival for Pancreatic…
Pancreatic cancer pill doubles life to 13 months By Tyler…
By – Tyler BrooksFDA warns public as cookie firm…
FDA warns public as cookie firm rejects urgent recall request…
By – Tyler BrooksTrump orders CDC to slash childhood…
Trump orders CDC to slash childhood vaccines from 17 to…
By – Tyler BrooksUSDA warns Americans over Salmonella in…
USDA warns Americans over Salmonella in meat products By Tylor…
By – Tyler BrooksGKN Aerospace’s Biggest Battle May Not…
By Samuel López | USA Herald The immediate danger of…
By – Samuel LopezGarden Grove Chemical Crisis Sparks Class…
By Samuel López | USA Herald A full-scale legal and…
By – Samuel LopezWembanyama in Tears: Spurs Dethrone Thunder…
Spurs dethrone Thunder in epic Game 7 road victory By…
By – Tyler BrooksSupreme Court signals 27 states could…
Supreme Court signals 27 states could ban trans female athletes…
By – Tyler BrooksMauricio Pochettino sounds alarm on Chris…
Mauricio Pochettino sounds alarm on Chris Richards injury By Tylor…
By – Tyler BrooksUSMNT star Chris Richards tears two…
USMNT star Chris Richards tears two ankle ligaments By Tylor…
By – Tyler Brooks“Money” Mayweather Tucks Tail: $100 Million…
Floyd Mayweather has beaten every opponent who ever climbed into…
By – Samuel LopezMackenzie Shirilla Sent Text Messages to…
Mackenzie Shirilla is once again at the center of public…
By – Jackie AllenNo posts found.
No posts found.