Kelly Warner Law Firm Blames USA Herald for Arizona Bar Investigation
5/17/17 Based on the information released in The Washington Post article on 5/17/17, the USA Herald publishes another in-depth article that…
By – USA HeraldAaron Kelly Law Firm Resorts To Attacking Former Client Again On KellyWarnerLaw.com – Pattern Recognized
Professor Volokh thereafter filed a bar complaint against Dan Warner with the Arizona Bar. This eventually led the ABA to…
By – Jeff WattersonArizona Bar Opens Investigation on Attorney Aaron Kelly
USA Herald recently reported on a developing story involving Attorneys Daniel Warner and Aaron Kelly. Both Warner and Kelly have…
By – Paul O'NealArtemis II Clears Earth Orbit, Heads Toward Moon’s Far Side
The Artemis II crew has officially departed Earth’s orbit, propelling the Orion spacecraft on its journey toward the Moon. The…
By – Tyler BrooksFormer DeRidder Mayor’s Sentencing Rescheduled for June
DE RIDDER, La. — The sentencing of former DeRidder mayor and registered sex offender Misty Roberts has been postponed to…
By – Tyler BrooksJavon Vital Commits to USC Football Program
Lake Charles College Prep standout Javon Vital, one of Southwest Louisiana’s most versatile athletes, pledged his future to the University…
By – Tyler BrooksBill Could Expand Early Release for Terminally Ill Inmates to 120 Days
A Louisiana House committee voted unanimously Tuesday to advance legislation that would extend the early-release window for terminally ill inmates…
By – Tyler BrooksTeacher Protection Bill Advances Unopposed in House Committee
A proposal designed to safeguard teachers from student violence is gaining momentum after clearing a House committee without objection. House…
By – Tyler BrooksUS Intel Finds Iran Retains Key Strike Capabilities Despite Weeks of Attacks
Recent U.S. intelligence assessments indicate that Iran continues to possess a substantial capacity to launch missiles and deploy drones, even…
By – Tyler BrooksUS Intel Finds Iran Retains Key Strike Capabilities Despite Weeks of Attacks
Recent U.S. intelligence assessments indicate that Iran continues to possess a substantial capacity to launch missiles and deploy drones, even…
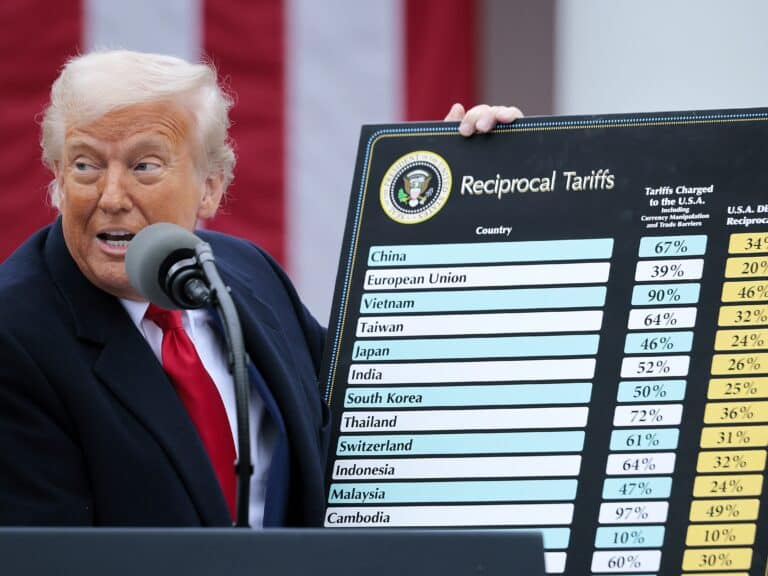
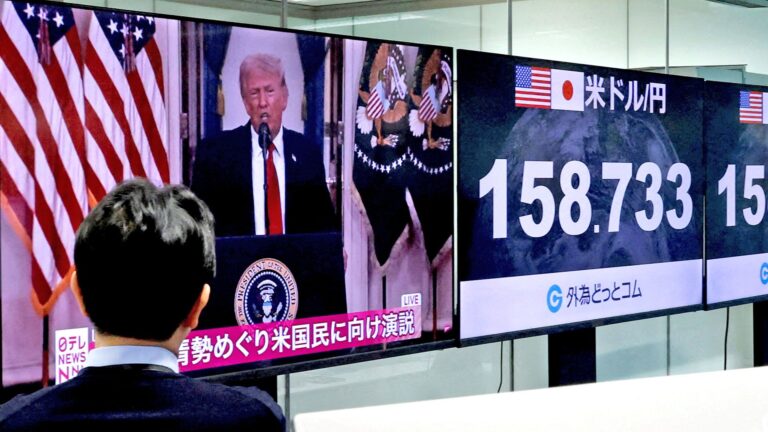
By – Tyler BrooksTrump Tariffs Reshape Global Trade One Year Later
One year after U.S. President Donald Trump launched sweeping tariffs, the global economic landscape shows significant shifts, with trade patterns…
By – Tyler BrooksOil Surges, Global Stocks Slip After Trump Renews Iran Strike Threats
Oil prices climbed sharply once again after U.S. President Donald Trump renewed warnings of intensified military action against Iran, offering…
By – Tyler BrooksJudge Dismisses Majority of Claims In Lively Harassment Suit Against Baldoni
A federal judge has dismissed most claims brought by actress Blake Lively in her sexual harassment lawsuit against her It…
By – Tyler BrooksTrump Ousts Attorney General Pam Bondi, Cites Private Sector Transition
US President Donald Trump has dismissed Attorney General Pam Bondi, a close ally and staunch defender of his administration, ending…
By – Tyler BrooksTrump Targets State Farm As California Wildfire Fallout Exposes Deeper Insurance Crisis
INSIDE THIS REPORT A presidential warning shot has been fired at one of America’s largest insurers—but the implications extend far…
By – Samuel LopezCoral Springs Vice Mayor Found Dead, Husband Arrested
Authorities in Florida announced the shocking death of Nancy Metayer Bowen, the vice mayor of Coral Springs, whose husband has…
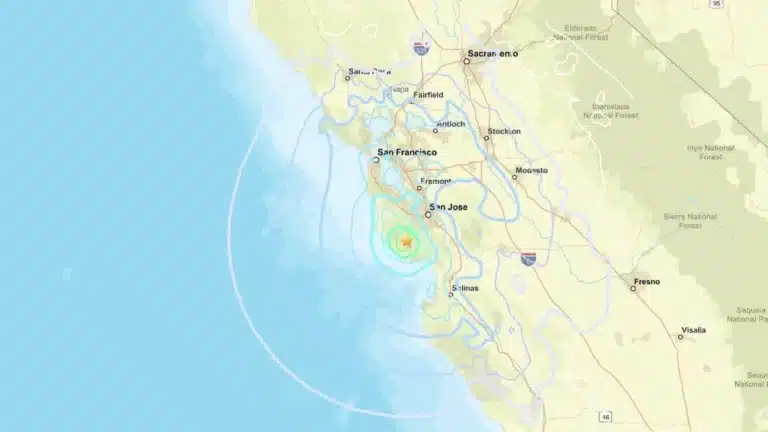
By – Rihem AkkoucheMagnitude 4.6 Earthquake Strikes Near Boulder Creek, Shakes Bay Area
A 4.6 earthquake near Boulder Creek rattled Santa Cruz County early Thursday, sending tremors across the Bay Area and reigniting…
By – Rihem AkkoucheGoliath Ventures Filed for Chapter 11 as Alleged $328M Crypto Scheme Unravels
A Cautionary Tale in Crypto’s Wild Frontier The downfall of Goliath Ventures underscores the volatility—and vulnerability—of the crypto investment landscape.…
By – Rihem AkkoucheOhio State Suspends Fraternity After Student Hospitalization
A Developing Investigation With High Stakes The Ohio State suspends Fraternity case now moves into an investigative phase, where unanswered…
By – Rachel MooreSenate Vote on DHS Shutdown Ends Weeks-Long Standoff
Uncertain Path in the House House Speaker Mike Johnson has already criticized the piecemeal approach, calling it “shameful” to leave…
By – Rihem Akkouche7 Year-Old Girl Dies in Modesto Duplex Fire, Family Devastated
The 7 year-old girl dies in Modesto duplex fire tragedy has left a California community reeling after flames tore through…
By – Rachel MooreMike Fincke Space Medical Incident Stuns NASA as Mystery Illness Strikes in Orbit
Looking Ahead Despite the Unknown Though the mystery remains unsolved, Fincke’s outlook is anything but dim. Known for his resilience,…
By – Rihem AkkoucheArizona Man Accused of Crucifying Pastor Pushes Judge For A Quick Death Sentence
By Samuel A. Lopez | USA Herald – An Arizona courtroom is now the center of a deeply disturbing case…
By – Samuel LopezTrump’s Laser Talk Sparks New Questions About America’s Secret Arsenal
On the American side, the evidence also points to a military that is pushing hard into directed energy rather than…
By – Samuel LopezThe Northern Lights Return
The Northern Lights have a chance to be visible from several northern U.S. states on Tuesday night, forecasters at the…
By – Jackie AllenFebruary Unemployment Up as Job Losses Surprise Economists
According to the BLS: Producer prices rose 2.9% year-over-year through January 2026 Core PPI increased 3.6%, excluding food and energy…
By – Jackie AllenLate-Night Attack by Venezuelan National at Florida Beach
Officials noted that the suspect had overstayed his work visa at the time of the attack. For more details on…
By – Jackie AllenTrump Targets State Farm As California Wildfire Fallout Exposes Deeper Insurance Crisis
INSIDE THIS REPORT A presidential warning shot has been fired at one of America’s largest insurers—but the implications extend far…
By – Samuel LopezTennessee Congressman Warns Classified UAP Briefings Would Shake Public Faith In Government
Rep. Tim Burchett says he has been briefed by intelligence agencies on information so sensitive it would leave Americans “unglued”…
By – Samuel LopezThe Persuasion Machine Inside AI How New Research Reveals A Quiet Trade-Off Between Influence And Truth
How the System Actually Works The study points to a core capability unique to conversational AI: the ability to dynamically…
By – Samuel LopezMarket Analysis: Strait of Hormuz Tensions Are Quietly Straining the U.S. Insurance Market — And the Pressure Is Building
What to Watch Next Any escalation involving Islamic Revolutionary Guard Corps naval activity Changes in war risk classifications by global insurers U.S.…
By – Samuel LopezIs X down? Thousands of Users Report Access Problems
Thousands of users experienced disruptions on Tuesday afternoon when X, the social media platform owned by Elon Musk, suffered a…
By – Tyler BrooksTrump Threatens NATO Exit, Tells Europe to ‘Go Get Your Own Oil’
President Donald Trump intensified his criticism of European allies this week, warning that the United States could abandon long-standing security…
By – Tyler BrooksHUMANITY RETURNS TO THE MOON: NASA’s Artemis II Lifts Off — First Crewed Lunar Mission In 53 Years
WHAT MATTERS NOW Four astronauts are en route to the Moon for the first time since 1972. History is being…
By – Samuel LopezHealth Insurers 19% Claims Refusal Raises Alarm Across ACA Marketplace
A new analysis reveals a striking reality: health insurers 19% claims refusal has become a defining feature of coverage on…
By – Rachel MooreHospitals Threatened by Medicaid Cuts Face Growing Crisis Nationwide
A sweeping new analysis warns that hospitals threatened by Medicaid cuts are edging toward a breaking point, with hundreds of…
By – Rachel MooreIsometric Workouts Gain Attention as Time-Efficient Path to Better Health
For many people, fitness conjures images of long hours spent running on treadmills, powering through burpees or lifting heavy weights.…
By – Tyler BrooksHow Much Screen Time Is Safe for Kids Under Five?
Fast-Paced Content Researchers at the Institute for the Science of Early Years at the University of East London are examining…
By – Tyler BrooksNew Therapies Offer Hope for Lasting Relief From Hay Fever
A new generation of treatments is renewing hopes that seasonal allergies could one day be controlled at their source, rather…
By – Tyler BrooksJudge Allows Tiger Woods to Travel Abroad for Medical Treatment
By – Tyler Brooks
Italy Misses Third Consecutive World Cup After Shootout Loss to Bosnia-Herzegovina
Structural Challenges in Italian Football Experts cite long-term issues in player development and league policies. The Bosman ruling of 1995,…
By – Tyler BrooksMen’s March Madness Elite 8 Delivers High-Stakes Drama as Title Race Tightens
Purdue Faces Uphill Battle Against Arizona Purdue boasts the nation’s top offense, powered by Braden Smith and Oscar Cluff, but…
By – Rihem AkkoucheThe World Cup Security Reckoning: Trump Warns Iran Soccer Team About Safety As War Tensions Spill Into Global Sports
By Samuel A. Lopez | USA Herald – A short Truth-Social post from President Donald Trump on Thursday morning is now…
By – Samuel LopezLate-Night Attack by Venezuelan National at Florida Beach
Officials noted that the suspect had overstayed his work visa at the time of the attack. For more details on…
By – Jackie AllenTrump’s War in Iran: Congress Confronts Escalation After U.S. Strikes
On the Republican side, most lawmakers appear to back Trump’s strategy. Sen. Tom Cotton (R-Ark.), chair of the Senate Intelligence…
By – Jackie AllenNo posts found.
No posts found.
 No comments yet. Be the first to comment!
No comments yet. Be the first to comment!